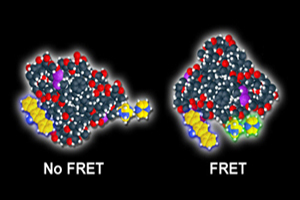
energy transfer (FRET) works. With FRET, the
illuminated yellow molecules come together,
signaling that they are transferring energy in the
living cell. (Image courtesy of Michael Davidson,
NHMFL)
Now that the genomes (DNA) of humans and many other organisms have been sequenced, biologists are turning their attention to discovering how the many thousands of structural and control genes—the “worker bees” of living cells that can turn genes on and off—function.
To do that, they need to develop new techniques and tools. Scientists in the Optical Microscopy group at the National High Magnetic Field Laboratory at Florida State University, working in collaboration with researchers from the University of Alberta in Canada and the University of California, San Diego, have done just that, and in the process have produced back-to-back articles in the prestigious journal Nature Methods.
In the first paper, magnet-lab biologists Michael Davidson and Kristin Hazelwood worked with researchers from the University of Alberta to create two new fluorescent-protein biosensors, molecular “beacons” that can tell if there is activity within a cell. The biosensors can be used simultaneously to monitor two separate dynamic functions in a single cell—a key to understanding how different proteins and enzymes (the biomolecules that cause chemical reactions) work together to complete the daily chores that help cells grow and divide. Knowing how cells work together can help researchers learn a great deal more about tumors and developmental biology, among many other things.

The researchers improved a powerful technique used to monitor cellular dynamics called fluorescence resonance energy transfer, or FRET. The technique is used to examine a new class of biosensor molecules that tether two fluorescent proteins together through an intervening peptide (which is like a polymer). Several hundred of these new biosensors have been developed over the past few years and are being used by scientists around the world to study a variety of functions, including programmed cell death, carbohydrate metabolism, cell division, hormone stimulation, acidity changes—just about any cellular process that can occur.
“In FRET, two molecules that are fluorescent act as ‘molecular beacons’ under the microscope, transferring energy between each other if they interact in the living cell,” said Davidson, who directs the magnet lab’s Optical Microscopy program. “With FRET, we can see that happen, but until now, we have only been able to monitor one biosensor at a time.”
The new technique, called Dual FRET, is outlined in the paper “Fluorescent Protein FRET Pairs for Ratiometric Imaging of Dual Biosensors.” It is available for purchase or via subscription at www.nature.com/nmeth/journal/v5/n5/abs/nmeth.1207.html.
Further expanding the capabilities of optical microscopy, Davidson and his team worked with collaborators from the University of California, San Diego to create a new screening method for fluorescent proteins that makes them more stable under the microscope. These proteins are sensitive to light, which can bleach them out after a certain period of time. By making the proteins more stable, microscopists can observe live cell dynamics for longer periods of time. The paper describing their work, “Improving the Photostability of Bright Monomeric Orange and Red Fluorescent Proteins,” was published in the May 4 online edition of Nature Methods. The paper is available for purchase or via subscription at www.nature.com/nmeth/journal/vaop/ncurrent/full/nmeth.1209.html.
Taken together, the new technique and tool are expected to speed up experiments and expand the utility of optical microscopy by allowing two dynamic processes inside a cell to be observed at once—and for longer periods of time.
The National High Magnetic Field Laboratory develops and operates state-of-the-art, high-magnetic-field facilities that faculty and visiting scientists and engineers use for research. The laboratory, which is operated by a consortium composed of Florida State University, the University of Florida and Los Alamos National Laboratory, is sponsored by the National Science Foundation and the state of Florida. To learn more, visit www.magnet.fsu.edu.




