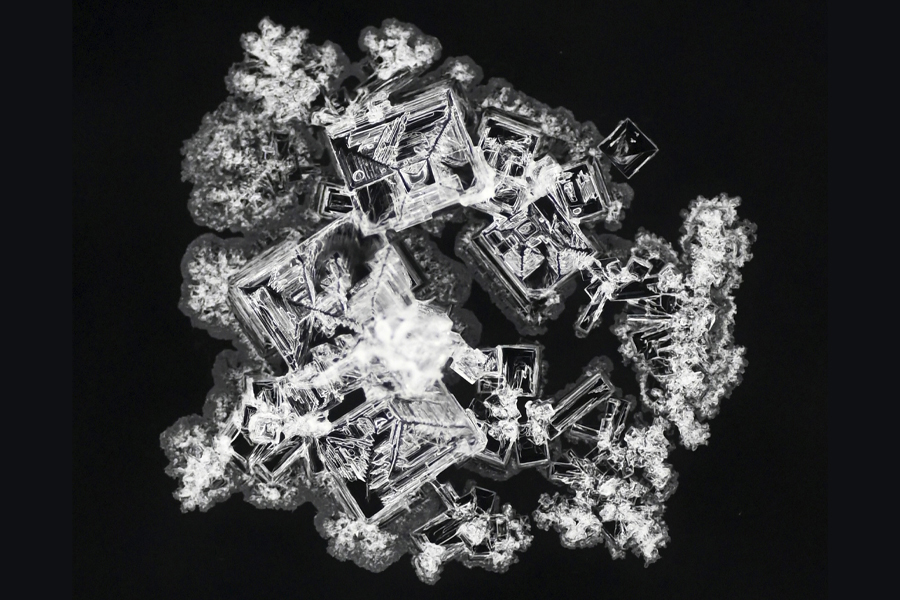
Imagine zooming in on a dried drop of salt solution — each pattern a unique masterpiece, reminiscent of abstract art, yet no larger than the size of a penny.

New research by scientists in the Florida State University Department of Chemistry and Biochemistry uses the patterns formed by a dried salt solution to train a machine learning algorithm that can identify the chemical composition of different salts. The work was published in Proceedings of the National Academy of Sciences.
“We are taking chemical fingerprints of different salts,” said Oliver Steinbock, Cottrell Family Professor of Chemistry. “Thinking of sodium chloride, or table salt, for example — among all samples of this type, they always look similar. There are differences from sample to sample, but all examples are distinct enough from other types that we can tell what kind of salt it is.”
When a salt solution dries, all sorts of forces are at play. Chemists have studied how fluid motion, crystal growth, environmental factors and other processes interact to determine the deposit pattern. The FSU researchers approached the problem from the opposite direction: If presented with a pattern of a dried solution droplet, could they determine what type of salt it was?
To do that, the researchers recorded 7,500 photos of 42 different types of salt stains. Using a new software approach, they translated each image into 16 parameters that can be rapidly analyzed by machine learning methods. The parameters capture features such as deposit area, compactness and texture. Each image was translated into numbers that in subtle ways encode the patterns’ arrangement of tiny crystals in rings, needles and leaf-like shapes.
To test their program’s ability to predict composition, the researchers analyzed additional images that were not part of the initial dataset. These programs successfully identified the correct salt in 90% of attempts.
“We were surprised at how well this worked,” Steinbock said. “Who would think that from a photo, you can tell the difference between sodium chloride and potassium chloride? They look very similar in the pictures. But the method is very good.”
The researchers plan to add to the training dataset by analyzing hundreds of thousands of new images, which would make their tool even more accurate and versatile. This vast number calls for lab automation, and such a robotic drop imager is currently being tested in Steinbock’s group.
“The difference between a human doing this and a computer is that the computer and our software can do this quantitatively,” Steinbock said. “If I were to present you 7,500 images, you would be confused. But the computer gets better and better with more and more images.”
The ability to quickly provide insight into the chemical composition of a sample from a photograph has many potential applications. For example, outfitting a rover exploring another planet with a full chemistry lab would be difficult and expensive, but a camera offers a cheap and lightweight alternative. Other scenarios, such as testing materials for lab safety, rapid screening for suspected drugs or low-cost blood analysis in places without access to hospitals, offer other potential applications.
Another benefit of this approach is that it only requires a minute amount of material. With just a few milligrams that make up a salt deposit, users could understand what they are likely handling and inform their decisions about how to proceed.
“If you want to have a rough idea of what that stain or spill is on a lab bench, you might use this as a cursory, first-step analysis,” said Bruno Batista, a senior researcher in Steinbock’s lab and the paper’s lead author.
Co-authors on this paper were Semhare Tekle, an undergraduate researcher in Steinbock’s lab; Jie Yan, a professor in the Department of Computer Science at Bowie State University; and Beni Dangi, an associate professor in the Department of Chemistry at Florida A&M University. This project was supported by NASA.
To see more images of salt patterns analyzed by Steinbock’s lab, visit the SaltScapes Gallery at his group’s website.






